Cancer, in all its ugly and mutinous forms, has proven to be a very challenging disease to cure. This is partly because cancer cells are often times difficult to target amongst healthy cells, and partly because they can mutate and evolve rapidly, which requires cancer treatments to similarly evolve under rigorous scientific scrutiny. And as if eliminating cancer cells wasn’t hard enough, growing research suggests that cancer cells can often evade existing treatments, sneaking past carefully crafted therapeutics and mutating into even more nefarious things.

Tim Hoffman, Ph.D.
Luckily, researchers in the Department of Biochemistry at the University of Colorado, Boulder are working to understand the particular problem of cancer cell “escapees” that aren’t destroyed by small molecule cancer therapeutics, and how to stop them from causing more damage to the body.
To learn more, Coffee Table Science interviewed Dr. Timothy Hoffman (TH), who led a study on cancer cells that survive drugs that are specifically used to target tumor growth via the mitogen-activated protein kinase pathway inhibitor (Don’t worry – we learned what this meant along the way, too).
CTS: Before delving into the meat of your study, I would love to know – what got you interested in studying cancer treatments in this capacity?
TH: My path to becoming a cancer biologist was certainly a winding one, but each of my subsequent experiences still built on each other in some way. I started out as a chemist many years ago in my undergrad studies, then I entered a PhD program in toxicology to understand how chemicals interact with and harm cells, then in my postdoctoral work I became really interested in how abnormal cancer cells are and how to effectively stop them from growing without harming normal cells. I feel that identifying newer and better cancer therapies is a great use of all my past experiences, and I see myself in this field for a very long time.
CTS: Your study focuses on cancer cells that escape certain small-molecule cancer therapeutics. What are small molecule cancer therapeutics, and how do they differ from typical cancer treatment?
TH: “Small molecule therapies” is actually a very wide umbrella term that includes typical cancer treatments. Older, more classical small molecule treatments include chemotherapies, which are very toxic to cancer cells and healthy cells. Newer, targeted small molecule therapies are much more refined, where the small molecules in this category are designed to exploit some distinct feature that only cancer cells possess, and thus primarily kill only the cancer cells.
CTS: These therapeutics target the mitogen-activated protein kinase (MAPK) pathway. What is the MAPK pathway, and what is its role in cancer growth?
TH: The MAPK pathway integrates many biological signals for a very wide array of cellular functions. One such function is to tell cells that it’s time to grow and perform cell division. Many cancers harbor a mutation in this pathway, and so they always have the “green light” signal to keep growing and keep dividing. Hence, the pathway has become a major target for therapeutic intervention.
CTS: Are the escapee cancer cells affected at all by MAPK inhibitor drugs? If so, is this a good thing or a bad thing for the patient?
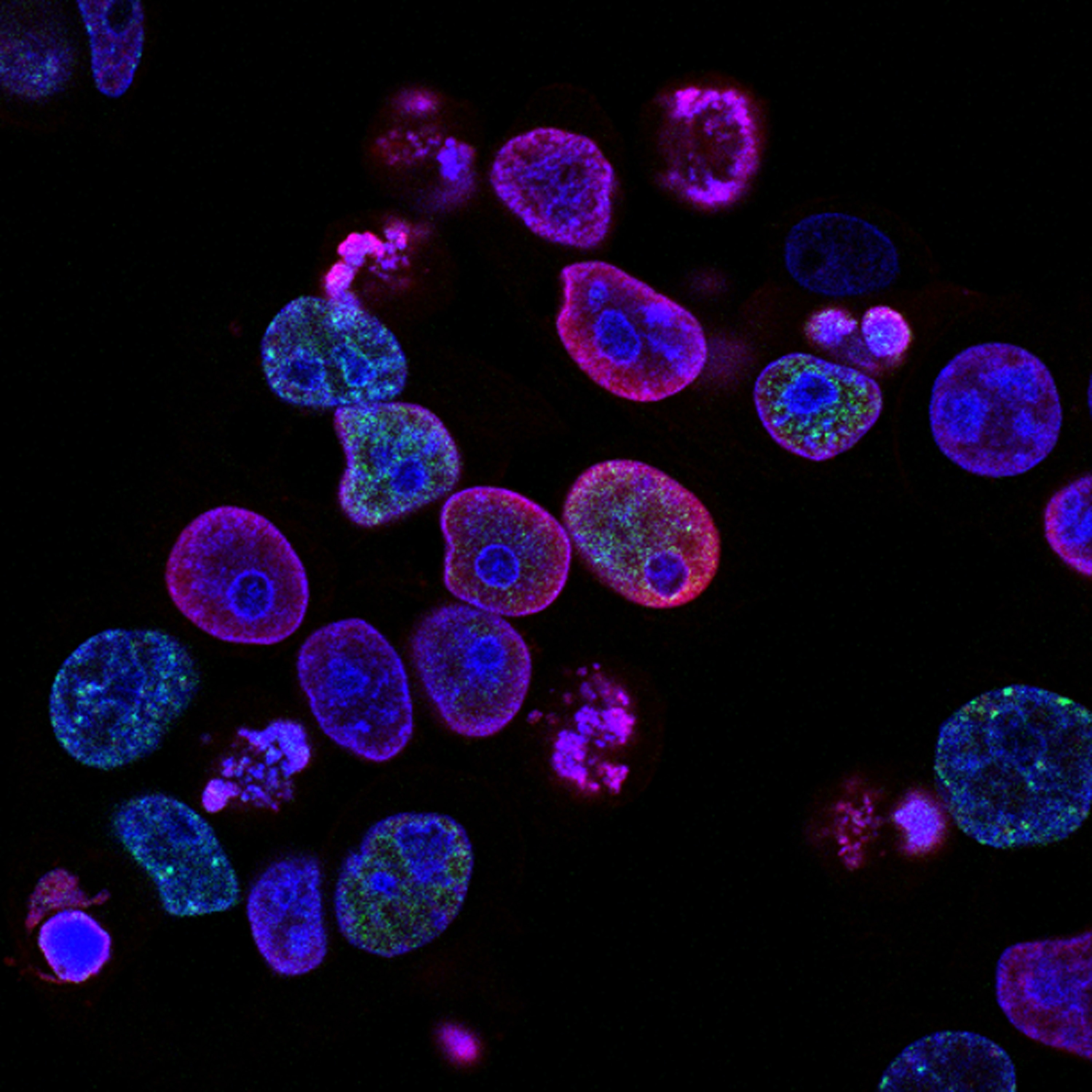
TH: MAPK inhibitors effectively target this pathway that the cancer cells are addicted to, but these escapees can adapt and figure out how to perform cell division anyway. Since their “green light” into the cell cycle looks different in this adapted state, it appears that they are not really prepared to replicate and divide, yet they proceed anyway. Collectively, this leads to some extra damage in the DNA that the cancer cells can tolerate, making them even more prone to cancerous evolution and potentially very harmful to the person.
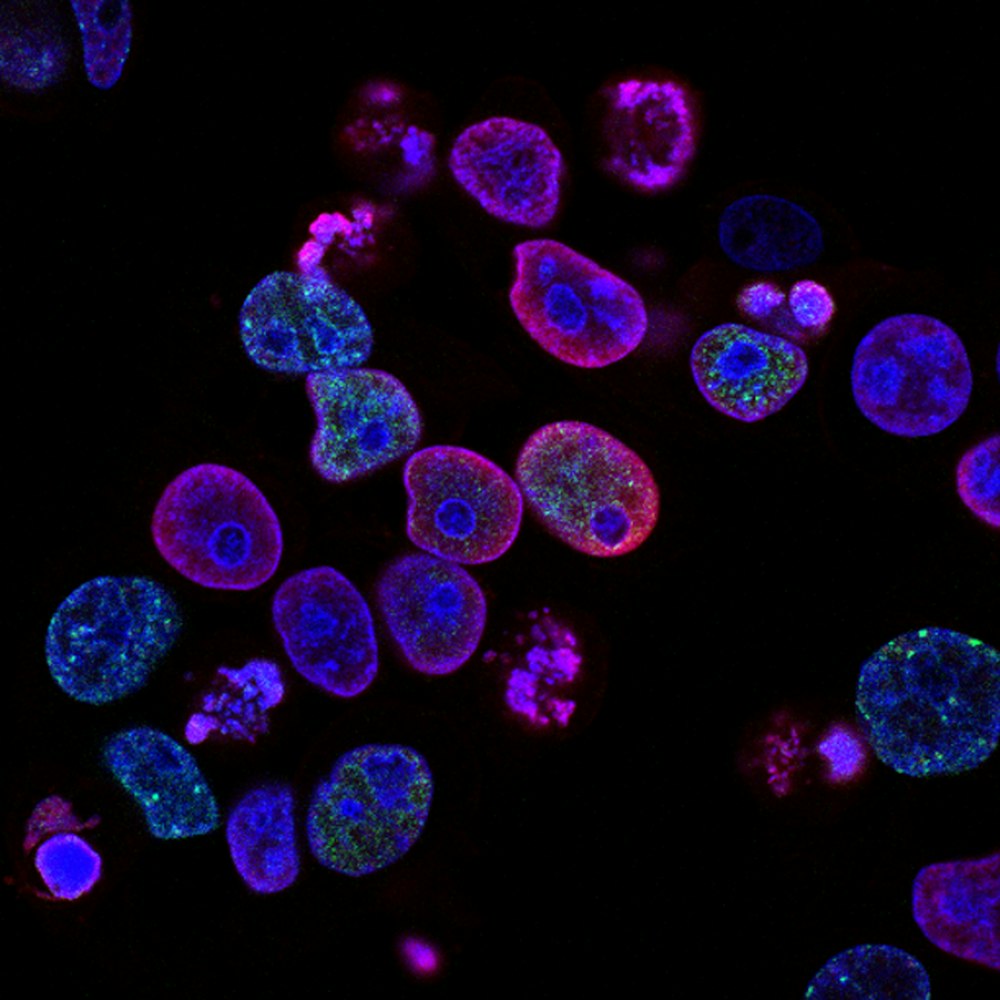
Image credit: Unsplash
CTS: One of your major conclusions was that targeting the Fanconi anemia (FA) pathway is a promising way to attack these escapee cells. Why do you think this is a good strategy?
TH: The Fanconi anemia (FA) pathway is a selective DNA repair pathway that these escapee cells seem to be broadly relying on to divide under these stressful treatments. It’s a fairly essential pathway for many cells, not just cancer cells, but short-term treatments targeting the FA pathway may be effective at wiping out the escapee cancer cells, and non-cancerous cells would hopefully be able to rebound after the treatment is removed. Even if that clinical application is not valid, at least this work adds to our understanding of cancer cell adaptation, and I hope that the knowledge can be used to develop other therapies that are more targeted toward killing cancer cells.
CTS: What sort of impact do you hope this work has on the field?
TH: I hope that this work will allow us as a community to broaden the scope of how we view drug resistance to targeted cancer treatments, as there may be many more similarities between drugs and cancer types than we realize or appreciate. Ultimately, my hope is that we can find some effective combination therapies that will be durable and curative for a very wide patient population.
The details of the research have been published in the journal Science.
If you enjoyed reading this post, please subscribe to our newsletter or Visit our Shop to buy some geeky science merchandise.


1 comment
I enjoy reading your posts regularly.